
Research Article
Volume-1 Issue-1, 2026
Novel Continuous Process of Emulsion Liquid Membrane for the Dysprosium Ion Extraction
-
Received Date: January 10, 2026
-
Accepted Date: January 25, 2026
-
Published Date: January 28, 2026
Journal Information
Switch to Full Text Menu
Abstract
In this work, for the first time, the efficiency of dysprosium (Dy) ion extraction by emulsion liquid membrane (ELM) in a continuous process has been investigated. The ELM consists of di-(2-ethylhexyl) phosphoric acid (D2EHPA) as extract ant, sorbitol monolete (span 80) as surfactant for stabilizing the emulsion phase, kerosene as diluent and nitric acid as stripping phase. The effects of operational parameters, such as pulsation intensity and flow rate of continuous and dispersed phases on efficiency of Dy extraction away from flooding point were studied. According to the results, increasing pulsation intensity leads to the enhancement of the efficiency of extraction. Furthermore, three empirical correlations for efficiency of extraction, dispersed phase mass transfer coefficient and dispersed phase Sherwood number are derived in terms of operating variables with a mean deviation of 0.29%, 14.135% and 11.33%, respectively. Moreover, the results showed that the favorable conditions obtained for the extraction process were at 4.78 L/h flow rate of continuous phase, 2.5 L/h flow rate of dispersed phase and 1.2 cm/s pulsation intensity. At these conditions, the maximum extraction of the Dy ion was 99.3%.
Key words
Emulsion Liquid Membrane, Continuous Process, Pulsed Packed Column, Dysprosium Ion, Mass Transfer Coefficient
| Material | Chemical formula | Density | Molecular mass | Surface tension | Stickiness |
| 970 | 322.43 | 20 | 52 |
| Surfactant | Chemical formula | Density | Molecular mass | Stickiness | |
| Span - 80 | 990 | 428.60 | 100-200 | 4.3 |
| Feed phase | Emulsion | Physical properties |
| 998 | 904.4 | )Density ) |
| 1.0 | 64.0 | () Stickiness |
| 11.o | Interphase tension | |
| Parameter | Values |
| 8 | Surfactant concentration |
| 0.05 | ()Carrier concentration |
| 4.0 | Feed phase ( ) |
| 1.0 | Internal phase concentration() |
| 50 | Dysprosium metal concentration in the feed phase () |
| Dispersed phase flow intensity () () | Continuous phase current intensity () () | The intensity of the impact () () | Test number |
| 1.5 | 2.3 | 0.8 | 1 |
| 1.5 | 2.3 | 1 | 2 |
| 1.5 | 2.3 | 1.2 | 3 |
| 1.5 | 3.7 | 0.8 | 4 |
| 1.5 | 3.5 | 1 | 5 |
| 1.5 | 3.7 | 1.2 | 6 |
| 1.5 | 5 | 0.8 | 7 |
| 1.5 | 5 | 1 | 8 |
| 1.5 | 5 | 1.2 | 9 |
| 2.3 | 2.3 | 0.8 | 10 |
| 2.3 | 2.3 | 1 | 11 |
| 2.3 | 2.3 | 1.2 | 12 |
| 2.3 | 3.7 | 0.8 | 13 |
| 2.3 | 3.7 | 1 | 14 |
| 2.3 | 3.7 | 1.2 | 15 |
| 2.3 | 5 | 0.8 | 16 |
| 2.3 | 5 | 1 | 17 |
| 2.3 | 5 | 1.2 | 18 |
| 3.7 | 2.3 | 0.8 | 19 |
| 3.7 | 2.3 | 1 | 20 |
| 3.7 | 2.3 | 1.2 | 21 |
| 3.7 | 3.7 | 0.8 | 22 |
| 3.7 | 3.7 | 1 | 23 |
| 3.7 | 3.7 | 1.2 | 24 |
| 3.5 | 5 | 0.8 | 25 |
| 3.7 | 5 | 1 | 26 |
| 3.7 | 5 | 1.2 | 27 |
| 0.055 | |
| 0.013- | |
| 0.014- | |
| -0.153 | |
| -0.007 | |
| 0.0003 | |
| 0.003 | |
| 0.001- | |
| 0.002 | |
| 0.078 |
| Percentage of relative error (__) | Predicted extraction yield ()(__) | Actual extraction yield (__)() | The final concentration of dysprosium in the feed phase ()() | The initial concentration of dysprosium in the feed phase ()() | Test number |
| 0.473 | 0.990 | 0.995 | 0.206 | 40.01 | 1 |
| 0.351 | 0.988 | 0.985 | 0.598 | 39.88 | 2 |
| 0.549 | 0.993 | 0.998 | 0.060 | 39.88 | 3 |
| 0.128 | 0.997 | 0.996 | 0.180 | 40.89 | 4 |
| 0.269 | 0.995 | 0.998 | 0.084 | 40.89 | 5 |
| 0.119 | 0.999 | 0.999 | 0.053 | 40.01 | 6 |
| 0.347 | 0.999 | 0.995 | 0.181 | 40.01 | 7 |
| 0.050 | 0.997 | 0.998 | 0.084 | 40.28 | 8 |
| 0.399 | 0.002 | 0.998 | 0.741 | 38.76 | 9 |
| 0.611 | 0.986 | 0.980 | 0.722 | 36.22 | 10 |
| 0.454 | 0.985 | 0.980 | 0.720 | 36.68 | 11 |
| 0.430 | 0.990 | 0.986 | 0.530 | 36.68 | 12 |
| 0.254 | 0.992 | 0.995 | 0.197 | 35.89 | 13 |
| 0.448 | 0.991 | 0.995 | 0.176 | 37.42 | 14 |
| 0.124 | 0.996 | 0.997 | 0.108 | 37.42 | 15 |
| 0.143 | 0.993 | 0.995 | 0.198 | 38.06 | 16 |
| 0.465 | 0.992 | 0.997 | 0.117 | 38.06 | 17 |
| 0.042 | 0.997 | 0.998 | 0.081 | 37.24 | 18 |
| 0.574 | 0.986 | 0.991 | 0.329 | 37.55 | 19 |
| 0.176 | 0.985 | 0.983 | 0.625 | 37.55 | 20 |
| 0.404 | 0.991 | 0.985 | O.200 | 39.02 | 21 |
| 0.282 | 0.990 | 0.987 | 0.489 | 38.86 | 22 |
| 0.230 | 0.990 | 0.988 | 0.482 | 38.73 | 23 |
| 0.340 | 0.996 | 0.982 | 0.296 | 38.37 | 24 |
| 0.082 | 0.990 | 0.990 | 0.399 | 38.27 | 25 |
| 0.036 | 0.990 | 0.990 | 0.392 | 38.11 | 26 |
| 0.165 | 0.996 | 0.998 | 0.091 | 38.11 | 27 |
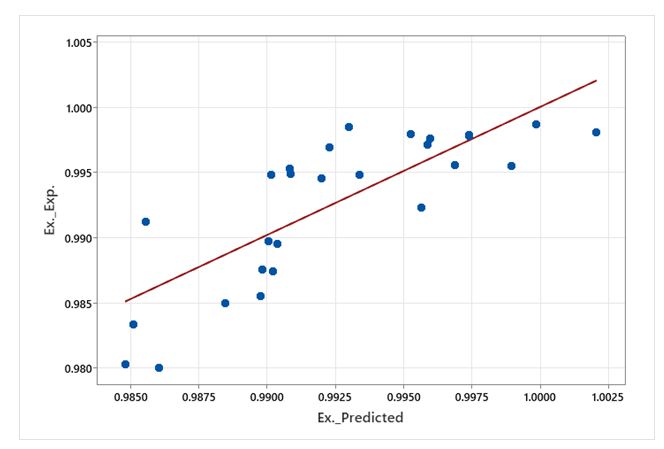 |
| Figure 1: Comparing the results obtained from the experimental and predicted relations of extraction efficiency |
 |
| Figure 2: The effect of impact intensity on the extraction efficiency in continuous phase flow intensity of 3.7 liters/hour and according to the intensity of dispersed phase flows of 1.5, 2.3 and 3.7 liters/hour(The concentration of span-80 as a material of surfactant is equal to 2.5 of the concentration of D2EHPA as an extracting ( ) is equal to 0.05, the feed phase is equal to 4, the concentration of the internal phase ( ) is equal to 1, and the concentration of dysprosium metal in the feed phase is equal to 50 ppm) |
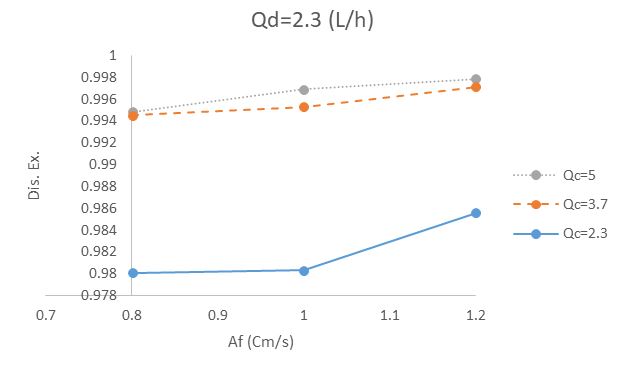 |
| Figure 3: The effect of impact intensity on the extraction efficiency in the flow intensity of the dispersed phase 2.3 liters/hour and for the flow intensity of the continuous phase 2.3, 3.7 and 5 liters/hour (The concentration of span-80 as a surfactant is equal to 2.5, the concentration of D2EHPA as an extracting ( ) is equal to 0.05, of feed phase is equal to 4, the concentration of the internal phase ( ) is equal to 1, and the concentration of dysprosium meta |
Introduction
Element of dysprosium (Dy) is a chemical material in the periodic table with atomic number 66. Dy in the 1950, with advances in techniques called ion exchange were isolated. It is a metal with a silver polish that is relatively stable at room temperature and dissolves in water [1-4]. This element is never found in nature as a free element, although it is found in the minerals monazite and bistanasite together with other earth elements [5-7]. It is also used in combination with vanadium and other elements in the manufacture of laser materials and commercial lighting. In nuclear industries, it is used as a neutron absorber in thermal reactor control rods. It is one of terfenol-D compounds along with iron and terbium [8-12]. It has the highest magnetic temperature of any known material and has various applications in advanced technologies such as converters, wide band mechanical resonators and injectors [13-20]. The main use of this element is in neodymium-iron-boron magnets, which are added to them in order to increase strength and corrosion resistance for required applications such as electric motors for electric vehicles and generators for wind turbines. Also, due to its high magnetic sensitivity, Dy is used in the field of data storage, which cannot be replaced by other chemical elements [21-25].
In liquid-liquid extraction, the feed and solvent phases are in contact with each other and the desired component is transferred from the feed phase to the solvent phase. It is used when the distillation process is not practical or expensive and has received much attention due to the need for materials with higher purity, the growing demand for temperature-sensitive products, and the use of solvents with high selectivity. The most common application of this method is related to petroleum industries, because in these industries, liquid feeds are separated based on their chemical species, and separation based on molecular weight or vapor pressure is not very useful [26-30]. Today, extraction processes are favored in order to separate and recover valuable metals or remove heavy metals that are found as pollutants in wastewater in various industries. Protraction includes the extraction of volatile and non-volatile organic compounds from liquids such as water with the help of a membrane. Protraction is classified as follows:
A) Without phase dispersion
1.Bulk liquid membrane
2. Stabilized liquid membrane
3. Liquid film protraction
B) With phase dispersion
1. Emulsion liquid membrane (ELM)
Protraction of ELM is effective for performing separation operations and since the use of membrane brings advantages such as simultaneous extraction and disposal operations, ease of operation, reduction of solvent consumption, low energy consumption and low operating costs, in the process separation and concentration of metal ions has received special attention. Some of the advantages of extracting metals using the liquid emulsion membrane method over other methods can be mentioned:
1. Very high specific surface area and consequently high mass transfer rate (30 times more than the fixed liquid membrane) [31].
2. The process of extraction and purification in the liquid emulsion membrane method is done in one step and simultaneously, and this means saving the volume of the equipment [32].
3. Selective transfer of one or more components from the external phase (feed phase) to the internal phase (discharge phase. The possibility of extraction from very dilute solutions [33].
Penetration in liquids is easier than in solids, so the liquid emulsion membrane process requires a lower mass transfer flux than using solid membranes. In other words, in order to compete with liquid membranes, polymer membranes must be less thick, which causes many operational problems [34].
Despite the mentioned advantages, this method faces two major challenges of swelling and emulsion failure. Swelling means the penetration of water from the external phase to the internal phase, which leads to thickening of the external phase and dilution of the internal phase. The failure usually occurs due to the surface cut between the membrane and the external phase. The emulsion liquid membrane process consists of 3 stages, which are as follows:
1. Preparation of emulsion membrane phase
2. The contact of the phases in order to carry out the extraction process (in this step, mass transfer from the aqueous phase to the organic phase takes place).
3. Breakdown of emulsions
In the first step, the internal phase prepared by dissolving nitric acid in water is poured into the membrane phase, which consists of extracting (carrier), surface active substance or emulsifier and diluent (solvent), in equal proportion and stirred. It turns into an emulsion. The emulsion consists of small droplets of the internal phase and is stable due to the presence of surface active substance (surfactant). In the second step, the emulsion comes into contact with the feed in the packed column and the goal is to transfer the mass from the feed phase. In this step, a double aqueous/organic/aqueous emulsion is obtained.
At this stage, by changing the operating conditions such as the flow intensity of the continuous and dispersed phases as well as the impact intensity, their effect on the amount of extraction is investigated. Also, in this step, in order to measure the diameter of the drop, a photo is taken from the active area of the column. Mass transfer in liquid-liquid systems is less than gas-liquid systems and the need for many theoretical steps is felt in order to increase mass transfer in extraction towers, so increasing the efficiency of liquid-liquid extraction columns is always a priority. To achieve this, mechanical energy can be used. One of the options is the use of impact extraction columns, the mechanical energy input to which is supplied through the input pulse to the tower. The input pulse plays the role of a mechanical stirrer and also produces droplets with smaller diameters, which increases the efficiency of the column in this case.
On the other hand, the volume of effluents in various industries is high, so it is better to use an ELM system in one of the extraction columns, such as a vertical shock column of the filled type. High capacity and efficiency in separation, insensitivity to two-phase interface pollution, relatively simple design, reliable and stable operation, no moving parts and suitable operation for radioactive and corrosive solutions, are among the prominent features of extraction towers. They are a blow. In the design of the column, parameters such as the average size of the dispersed phase droplets, the amount of the dispersed phase, and the mass transfer coefficient should be determined.
The amount of dispersed phase is determined by quenching method and the average size of dispersed phase droplets is determined by photographing the dispersed phase in the column. Also, to calculate the mass transfer coefficient, it is written around the mass balance column. Impact extraction columns have many uses in chemical processes. The best application of this type of towers is in the nuclear fuel industry, which has an advantage over other mechanical contractors due to its reliable operation and remote control capability.
In addition to the nuclear industry, in many chemical industries, extraction, which is an important process in chemical engineering operations, is used. Impact towers are used in three types: tray impact towers, filled impact towers and disk-donut impact towers [35-39].
Considering the advantages of percolation extraction compared to conventional liquid-liquid extraction methods, this method can be the introduction to the use of ELM for the separation of various elements with a very small concentration (less than 100) in packed columns on an industrial and semi-industrial scale. In fact, one of the important innovations of this research is the use of ELM in a stably filled vertical shock column, the examination of the dependence of the extraction efficiency on the operating parameters, and also the possibility of replacing ELM method instead of the conventional liquid-liquid extraction in it is a packed vertical impact column, which is suggested to use ELM in packed impact towers due to high extraction efficiency. It is also worth noting that ELM has many advantages compared to traditional methods due to the reduction of some process steps from the environmental and economic point of view.
Experimental and Method
In ELM protraction, the carrier is one of the important factors that play an important role in the mass transfer process and its price is also high. In these experiments, a strong acid carrier or extracting called span-80 was used as a surfactant. It shows the best physical properties of the carrier and surfactant. It refers to the balance of hydrophilicity and lipophilicity of the surfactant, they are presented in the Tables 1 and 2.
By dissolving dysprosium metal salt in distilled water, the feed phase solution was obtained. In order to reduce the solubilization error, first, 1000 solution was prepared and then the feed solution used in the experiments, i.e. solutions of 50 ppm, was obtained by diluting the concentrated solution. It was also measured of pH, the feed phase and adjusted to number 4 by adding nitric acid.
Internal phase solution is prepared by dissolving a certain amount of nitric acid, which is a stripping agent, in distilled water. In below, the values of the optimal parameters used in these experiments in the production of emulsion liquid membrane and feed solution are given, and these values are the optimal results on the extraction of dysprosium by emulsion liquid membrane in a discontinuous system. The purpose of this stage is to make emulsion consisting of aqueous phase in organic phase. The ratio of internal phase to membrane phase is one to one. First, the cruzen, which is already well saturated with water, is poured into a special container and placed under the mixer, and span-80 as a surfactant with a concentration of 2.5% by volume is added to the cruzen and the mixer is turned on. Time is given for the surfactant to dissolve in cruzen. Also, in this step, the carrier is added to the solution with a certain concentration. Then, the internal phase is added drop by drop to the mixture and the stirring of the mixture continues at 12,000 rpm for 20 minutes. This step is one of the most important steps because the preparation of an emulsion with a long stability period that increases the extraction efficiency depends to a large extent on this step. If the emulsion is not stable at the time of entering the filled vertical shock extraction column, it will cause a lot of problems and the mass transfer operation will also be completely problematic.
Therefore, the correct formulation of the membrane as well as high stability are very important. At the end of this stage, the emulsion consisting of the aqueous phase in the organic phase or the dispersed phase is obtained, in which the volume ratio of the internal aqueous phase to the organic phase or membrane is equal to 1. The following table shows the physical properties of the used system, they are presented in the Tables 3 and 4.
The tests were carried out in different operating conditions and will be accompanied by mass transfer. The tests were carried out in the range of optimal operating conditions, scattered phase flow intensity (1.5, 2.3, 3.7) liters per hour, continuous phase flow intensity (2.3, 3.7, 5) liters per hour and impact intensity (0.8, 1, 1.2) cm/s were performed. In this way, 27 tests were performed below the overflow point. In fact, these values of the operational parameters used were chosen in such a way that the tests were performed below the overflow point. Analysis was used to check dysprosium concentration after extraction.
Results and Discussion
In this research, ELM composed of D2EHPA as a carrier, cruzen as a diluent and nitric acid as a stripping agent is used in a packed vertical multiplication column, in which dysprosium is transferred as a component from the feed phase that enters from the top of the column into the internal phase that it is entered from the bottom of the column.
First, using the minitab software, a relation for the extraction efficiency, which is a graph of the amount of dysprosium transferred from the feed phase to the membrane phase, was presented and its error was calculated. Then, the effect of operating parameters such as flow intensity of continuous and dispersed phases and impact intensity and the effect of each of them on the extraction efficiency was investigated. The effect of hydrodynamic factors such as the average diameter of the dispersed phase droplets and the amount of dispersed phase, which influence the design and enlargement of the filled multiplication columns, was expressed on the extraction efficiency. In the following, with the help of minitab software, the optimal mode was obtained to obtain the maximum extraction efficiency and the minimum intensity of the dispersed phase flow.
In addition, by writing the overall mass balance around the column, a relation was found for the mass transfer coefficient and a comparison was made between the results obtained and the methods of Newman, Kroenig, Brink, Handels and Baron. Also, with the help of dimensional analysis and using Minitab software, a relation for the mass transfer coefficient and a relation for the Sherwood number were obtained in terms of operational variables, and the error of these two relations was calculated and compared with each other. The range of values of the mentioned parameters have been selected keeping in mind the flood conditions so that all the tests are performed below the flood point. The operating conditions of the tests are given in the Table 5.
ws the initial and final concentration of Dy in the feed phase (donor phase), the actual extraction efficiency and the predicted extraction efficiency, and the relative error percentage of each experiment. The relative error means the result of dividing the difference between the actual and predicted extraction efficiency by the actual extraction efficiency. Equation 1-4 is considered for the extraction efficiency, in which the individual and interactive effects of operational parameters on the extraction efficiency are considered.
In relation 1-4, the flow intensity of the continuous and dispersed phases is in liters per hour and the impact intensity is in centimeters per second. The constants obtained from fitting the operational parameters with the help of minitab software are given in the Table 6.
Ignoring very small coefficients k1 and k2the following relationship is obtained:
Using equation 3-4, it is possible to calculate the average absolute relative error percentage of the predicted relation for extraction percentage compared to its experimental value:
In relation 3-4, N is the number of tests.
Based on the fixed coefficients presented, the average absolute relative error percentage in the predicted relationship for the extraction yield was obtained as 0.29, it is presented in Tables 7.
Figure 1 shows the comparison of the results obtained from the experimental and predicted relationship of extraction efficiency.
Figure 2 shows the effect of impact intensity on extraction efficiency in the case of constant flow intensity of the continuous phase.
In these experiments, in order to avoid flooding conditions, operational parameters were chosen with great care. In the range considered for the impact, there was no flooding in any of the tests, but it should also be noted that an excessive increase in the intensity of the impact brought the column into the unstable operational area, which corresponds to the occurrence of flooding and the inventory The dispersed phase increases, resulting in a decrease in efficiency. In the test conditions, increasing the intensity of the impact causes an increase in the flow rate and a decrease in the diameter of the dispersed phase droplets, hence the mass transfer level and as a result the mass transfer rate from the continuous phase to the dispersed phase also increases.
Figure 3 shows the effect of impact intensity on extraction efficiency in the case of constant dispersed phase current intensity.
In this case, the increase in the intensity of the impact leads to an increase in the flow turbulence and a decrease in the diameter of the dispersed phase droplets, so the mass transfer level and as a result the mass transfer rate increases. Increasing the intensity of the continuous phase flow prevents the rapid upward movement of the dispersed phase droplets and causes the droplet stay time in the column to increase.
On the other hand, this resistance against the upward movement of the dispersed phase can have a two-sided effect on the extraction efficiency. For example, for the constant current intensity of the continuous phase, if the current intensity of the dispersed phase is very high, as mentioned, the continuous phase plays a resistance role and increases the extraction efficiency, but if the current intensity of the dispersed phase is small, the other continuous phase plays a resistance role. did not play, and on the contrary, its large amount can have a negative effect on the distribution of dispersed phase droplets and reduce the extraction efficiency. As the intensity of dispersed phase flow increases, the ascent speed of the droplets increases, due to which the mass transfer level decreases and the droplets have less opportunity to transfer mass. On the other hand, increasing the flow intensity of the dispersed phase means increasing the emulsion and receiver phase. Therefore, according to the test conditions and the interval considered for the operating parameters, the exact effect of the flow intensity of the continuous and dispersed phases on the extraction efficiency should be studied. It should be noted that avoiding flood conditions is a decisive factor in choosing the right interval for operating parameters. In this work, in order to avoid flood conditions, a small range was considered for continuous phase current intensity changes.
Conclusion
The development of continuous process of emulsion liquid membrane can be very beneficial from the environmental and economic point of view. Using minitab software, a relationship for extraction efficiency was expressed in terms of operational parameters such as flow intensity of continuous and dispersed phases and impact intensity, and the average absolute relative error percentage of the predicted relationship for extraction efficiency was determined to be 0.29. The extraction efficiency from the external phase to the membrane phase in optimal conditions was 99.3%. The effect of operating parameters on extraction efficiency was studied, and the results were as follows:
1. In the case of constant continuous phase flow intensity, increasing the impact intensity causes an increase in flow turbulence and a decrease in the diameter of the dispersed phase droplets, hence the mass transfer level and as a result, the mass transfer rate from the continuous phase to the dispersed phase also increases.
2. In the case of constant dispersed phase flow intensity, the impact intensity increases, the flow turbulence increases and the diameter of the dispersed phase droplets decreases, so the mass transfer level and as a result the mass transfer rate from the continuous phase to the dispersed phase increases.
By examining the influence of different parameters on the efficiency of dysprosium extraction, the results indicated that in the test conditions and the range considered for operational parameters, the extraction efficiency is significantly greater than the flow intensity of the continuous and dispersed phases and to a lesser extent than the intensity The impact was effective. In the investigated conditions, the effect of continuous phase flow intensity was 54.33%, also, dispersed phase flow was 30.88% and impact intensity was 14.79% on extraction efficiency. Also, the effect of hydrodynamic factors such as the average diameter of the dispersed phase droplets and the amount of dispersed phase, which influence the design and enlargement of the filled columns, on the extraction efficiency was stated, and the results are as follows:
1. By reducing the droplet diameter, the extraction efficiency increases. In fact, by decreasing the droplet diameter, the mass transfer surface increases and as a result, the mass transfer rate also increases.
2. It is clear that the extraction efficiency decreases with the increase in the amount of dispersed phase in the column.
In the liquid emulsion membrane process, in order for the separation process to be economical, optimization is done to minimize the carrier concentration, because the carrier is more expensive compared to other materials. In this column, the optimal state corresponding to the maximum level for the extraction percentage and the minimum level for the intensity of the dispersed phase flow, which is a graph of the amount of emulsion used, was considered. In the optimal conditions obtained by minitab software, the continuous phase flow intensity is 4.78 L/h, the dispersed phase flow intensity is 2.5 L/h, the impact intensity is 1.2 cm/second and the extraction efficiency is 99.3%.
Dimensionless groups were determined with the help of dimensional analysis and the relationship between operating parameters and the mass transfer coefficient of the dispersed phase was obtained using minitab software. The average absolute relative error percentage of the predicted relationship for the mass transfer coefficient of the dispersed phase was 14.135. The mass transfer coefficient of the dispersed phase was calculated with the help of Newman, Kroenig and Brink and Handels and Baron methods, and the average absolute relative error percentage of each of them was 32.6, 90.4 and 21.67 compared to the actual value of the experiments. With the help of dimensional analysis, dimensionless groups were determined and using minitab software, a relationship between Reynolds and Schmidt numbers and Sherwood number of dispersed phase was obtained. The average absolute relative error percentage of the predicted relationship for the Sherwood number of the dispersed phase was 11.33.
Using the values related to the Sherwood number of the dispersed phase which were calculated by the predicted equation, the equivalent values of the mass transfer coefficient of the dispersed phase were obtained and it was observed that in this case the percentage of error calculated for the mass transfer coefficient of the dispersed phase was up to 11.25 decreased. Therefore, the accuracy of the relationship predicted for the Sherwood number is higher than the accuracy of the relationship predicted for the mass transfer coefficient, and it is better to use the relation presented for the Sherwood number to calculate the mass transfer coefficient.
References
- Benderrag A, et al (2019) Experimental and modeling studies on Cd (II) ions extraction by emulsion liquid membrane using Triton X-100 as biodegradable surfactant. Journal of environmental chemical engineering, 7: 103166.
- Davoodi-Nasab P, et al. (2018) Selective separation and enrichment of neodymium and gadolinium by emulsion liquid membrane using a novel extractant CYANEX® 572. Minerals Engineering, 117: 63-73.
- Noah NFM, et al. (2016) Highly selective transport of palladium from electroplating wastewater using emulsion liquid membrane process. Journal of the Taiwan Institute of Chemical Engineers, 64: 134-41.
- Zaheri P, R Davarkhah (2017) Rapid removal of uranium from aqueous solution by emulsion liquid membrane containing thenoyltrifluoroacetone. Journal of environmental chemical engineering, 5: 4064-8.
- Daraei P, et al. (2019) Application of nontoxic green emulsion liquid membrane prepared by sunflower oil for water decolorization: process optimization by response surface methodology. Journal of Industrial and Engineering Chemistry, 77: 215-22.
- Davoodi-Nasab P, et al. (2018) Evaluation of the emulsion liquid membrane performance on the removal of gadolinium from acidic solutions. Journal of Molecular Liquids, 262: 97-103.
- Hachemaoui A, K Belhamel (2017) Simultaneous extraction and separation of cobalt and nickel from chloride solution through emulsion liquid membrane using Cyanex 301 as extractant. International Journal of Mineral Processing, 161: 7-12.
- Razo-Lazcano TA et al. (2018) Chlorpheniramine recovery from aqueous solutions by emulsion liquid membranes using soy lecithin as carrier. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 536: 68-73.
- Srivastava A, et al. (2017) Comparative study of arsenic (V) removal from aqueous solution using Aliquat-336 and 2-ethyl hexanol through emulsion liquid membrane. Journal of water process engineering 16: 64-8.
- Mokhtari B, K Pourabdollah (2015) Emulsion liquid membrane for selective extraction of Bi (III)." Chinese Journal of Chemical Engineering, 23: 641-5.
- Sulaiman RNR et al. (2014) Emulsion liquid membrane stability in the extraction of ionized nanosilver from wash water. Journal of Industrial and Engineering Chemistry, 20: 3243-50.
- Balasubramanian A, S Venkatesan (2012) Removal of phenolic compounds from aqueous solutions by emulsion liquid membrane containing Ionic Liquid [BMIM]+[PF6]− in Tributyl phosphate. Desalination, 289: 27-34.
- Seifollahi Z, A Rahbar-Kelishami (2017) Diclofenac extraction from aqueous solution by an emulsion liquid membrane: parameter study and optimization using the response surface methodology." Journal of Molecular Liquids, 231: 1-10.
- Kulkarni SS et al. (2018) Intensification of emulsion liquid membrane extraction of uranium (VI) by replacing nitric acid with sodium nitrate solution. Chemical Engineering and Processing-Process Intensification, 125: 18-26.
- Ng YS, et al. (2010) Performance evaluation of organic emulsion liquid membrane on phenol removal." Journal of hazardous materials, 184: 255-60.
- Kumbasar RA (2010) Selective extraction of chromium (VI) from multicomponent acidic solutions by emulsion liquid membranes using tributhylphosphate as carrier. Journal of hazardous materials, 178: 875-82.
- Raji M, et al. (2018) Selective extraction of dysprosium from acidic solutions containing dysprosium and neodymium through emulsion liquid membrane by Cyanex 572 as carrier. Journal of Molecular Liquids, 254: 108-19.
- Raji M, et al. (2017) Pertraction of dysprosium from nitrate medium by emulsion liquid membrane containing mixed surfactant system. Chemical Engineering and Processing-Process Intensification, 120: 184-94.
- Anitha M, et al. (2015) Extraction of neodymium from nitric acid feed solutions using an emulsion liquid membrane containing TOPO and DNPPA as the carrier extractants. Chemical Engineering Research and Design, 98: 89-95.
- Liu H, et al. (2017) Optimization of vanadium (IV) extraction from stone coal leaching solution by emulsion liquid membrane using response surface methodology. Chemical Engineering Research and Design, 123: 111-9.
- Boyadzhiev L, Z Lazarova (1995) Liquid membranes (liquid pertraction). Membrane Science and Technology, Elsevier, 2: 283-352.
- Kumar A, S Hartland (1999) Correlations for prediction of mass transfer coefficients in single drop systems and liquid–liquid extraction columns. Chemical Engineering Research and Design 77: 372-84.
- Pacek A, et al. (1998) On the Sauter mean diameter and size distributions in turbulent liquid/liquid dispersions in a stirred vessel. Chemical Engineering Science, 53: 2005-11.
- Pabby AK, et al. (2008) Handbook of membrane separations: chemical, pharmaceutical, food, and biotechnological applications, CRC press.
- Zereshki S, et al. (2021) Application of a green emulsion liquid membrane for removing copper from contaminated aqueous solution: Extraction, stability, and breakage study using response surface methodology. Journal of Molecular Liquids, 325: 115251.
- Othman N, et al. (2006) Separation of silver from photographic wastes by emulsion liquid membrane system." Journal of membrane science, 282: 171-7.
- Baker RW (2012) Membrane technology and applications, John Wiley & Sons.
- Davoodi-Nasab P, et al. (2019) Simultaneous effect of nanoparticles and surfactant on emulsion liquid membrane: Swelling, breakage and mean drop size." Separation and Purification Technology, 219: 150-8.
- Zereshki S, et al. (2018) Application of edible paraffin oil for cationic dye removal from water using emulsion liquid membrane. Journal of hazardous materials, 356: 1-8.
- Cahn R, N Li (1974) Separation of phenol from waste water by the liquid membrane technique." Separation Science, 9: 505-19.
- Kislik VS (2009) Liquid membranes: principles and applications in chemical separations and wastewater treatment, Elsevier.
- Volesky B (2001) Detoxification of metal-bearing effluents: biosorption for the next century." Hydrometallurgy, 59: 203-16.
- Araki T, H Tsukube (1990) Liquid membranes: chemical applications, CRC press.
- Kentish S, G Stevens (2001) "Innovations in separations technology for the recycling and re-use of liquid waste streams." Chemical Engineering Journal, 84: 149-59.
- Chen J, et al. (2002) Measurement of interface level, holdup, pulsation frequency, and amplitude in a pulsed column by air purge. Industrial & engineering chemistry research, 41: 1868-72.
- Moris MA, et al. (1997) Hydrodynamics of a rotating disc contactor. Separation and Purification Technology, 11: 79-92.
- Bahmanyar H, et al. (2008) Prediction of effective diffusivity and using of it in designing pulsed sieve plate extraction columns. Chemical Engineering and Processing: Process Intensification, 47: 57-65.
- Patel JM (1966) Mass transfer in droplets with turbulent internal circulation-Mathematical description.
- Mirmohammadi SL, Safdari J, Mallah MH (2023) Using optimal squared-off cascades instead of optimal square cascades to produce enriched uranium required for fresh fuel in the equilibrium cycle in power reactors. Annals of Nuclear Energy, 192: 109987.
Article Information
Research Article
Received Date: January 10, 2026
Accepted Date: January 25, 2026
Published Date: January 28, 2026
Novel Continuous Process of Emulsion Liquid Membrane for the Dysprosium Ion Extraction
Volume 1 | Issue 1
Citation
Mohammad Hassan Mallah, Jaber Safdari, Hossain Abolghasemi, Ali Soleiman Fallah (2026) Novel Continuous Process of Emulsion Liquid Membrane for the Dysprosium Ion Extraction. J Chem Reac Catal Res 1:102
Copyright
©2026 Mohammad Hassan Mallah. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

doi: jcrc.2026.1.102

