
Research Article
Volume-1 Issue-2, 2025
Sickle Cell and Salmonella: A Hidden Health Threat for Senegalese Children
Received Date: September 03, 2025
Accepted Date: September 16, 2025
Published Date: September 20, 2025
Journal Information
Abstract
Background: Infections are among the most serious complications of Sickle cell disease, especially in children under five years of age, who are at heightened risk due to functional asplenia and immune dysregulation. This study aimed to characterize the burden of infections in this high-risk group.
Methods: A retrospective review of physical medical records was conducted for all children and adolescents diagnosed with SCD who received care between January and December 2024. Data were extracted from hospital registries and included demographic information, hemoglobin electrophoresis profiles, reasons for consultation, identified pathogens, and results of antibiotic susceptibility testing.
Results: Of the 240 SCD patients included, 56 (23.3%) presented with infectious complications. Females accounted for 55.36% of these cases. The majority of infections occurred in children under 10 years, with 53.6% in the 0–5 years age group and 35.7% in the 5–10 years group. Septicemia was the most frequent infection (37%), followed by osteomyelitis (25%) and bronchopneumonia (20%). Less common infections included urinary tract infections (9%), arthritis (5%), and myositis (4%). Salmonella spp. was the predominant pathogen (29%), followed by Staphylococcus aureus (16%), Escherichia coli and Streptococcus pneumoniae (14% each). Antibiotic susceptibility testing revealed that Salmonella spp. strains were fully sensitive to all tested antibiotics, including Amoxicillin, Ertapenem, and Cotrimoxazole.
Conclusion: Invasive bacterial infections, particularly septicemia and osteomyelitis, are prevalent among pediatric SCD patients in Senegal. Salmonella spp. emerged as the leading pathogen, with encouraging antibiotic sensitivity profiles. These findings underscore the need for targeted infection prevention strategies and optimized antimicrobial stewardship in this vulnerable population.
Key words
Sickle Cell Disease, Salmonella Spp. Septicemia, Escherichia, Pneumonia, Cotrimoxazole
| Sex | 0–5 years | 5–10 years | 10–15 years | 15–20 years | Total |
| Male | 13 (52%) | 9 (36%) | 2 (8%) | 1 (4%) | 25 (44.64%) |
| Female | 17 (54.8%) | 11 (35.5%) | 3 (9.7%) | 0 | 31 (55.36%) |
| Total | 30 (53.6%) | 20 (35.7%) | 5 (8.9%) | 1 (1.8%) | 56 (100%) |
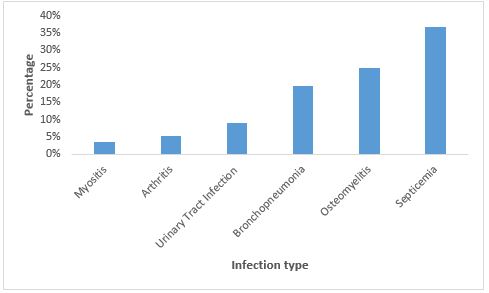 |
| Figure 1: Distribution of Infection Types Among Children with Sickle Cell Disease |
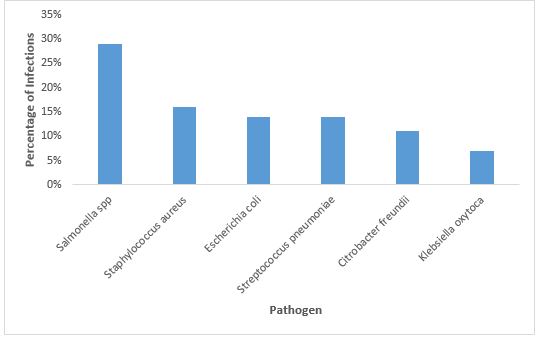 |
| Figure 2: Distribution of Infections by Pathogen in Children with Sickle Cell Disease |
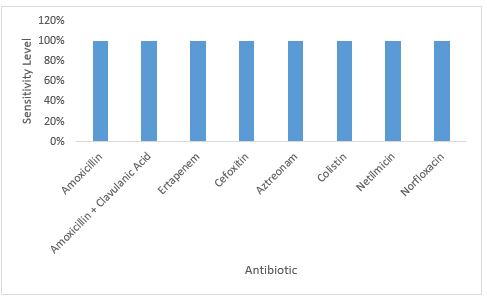 |
| Figure 3: Antibiotic Susceptibility profile of Salmonella spp. in Children with Sickle Cell Disease |
Introduction
Sickle Cell Disease (SCD) is a group of inherited blood disorders characterized by the presence of abnormal hemoglobin, which impairs the ability of red blood cells to transport oxygen efficiently throughout the body. The most severe form, sickle cell anemia, is a major global public health concern due to its high morbidity and mortality, particularly in low- and middle-income countries [1].
Africa bears the highest burden of SCD, accounting for nearly 80% of global cases, with carrier prevalence ranging from 2% to 30% depending on the region, with approximately 8 to 10% of Senegalese being carriers [1,2]. The natural history of sickle cell anemia is marked by chronic hemolytic anemia beginning in infancy, often accompanied by vaso-occlusive crises (VOCs), which can lead to acute and chronic complications, including organ damage and disability. One of the most critical challenges in managing SCD is the high susceptibility to infections, particularly among children under five years of age [1]. In sub-Saharan Africa, infections remain a leading cause of early mortality among children with SCD. According to the World Health Organization (WHO), SCD was responsible for over 81,000 deaths in children under five in 2021, making it one of the top causes of mortality in this age group [1].
Despite the availability of preventive measures such as vaccination and antibiotic prophylaxis, infectious diseases continue to pose a serious threat to this vulnerable population [3,4]. A study conducted in Libreville, Gabon, demonstrated that infectious diseases remain common and can be fatal among children with sickle cell disease (SCD) [5]. In Senegal, although data on infection rates in this population are limited, a retrospective study at Albert Royer Children’s Hospital in Dakar reported that 107 out of 327 children with SCD admitted to the emergency department (32.7%) presented with infections [6].
These findings highlight the ongoing vulnerability of children with SCD to infectious complications and underscore the need for further research to better characterize the burden of infections in this high-risk group.
Methodology
Study Design and Setting
We conducted a retrospective study based on physical medical records collected between January and December 2024 at Diamniadio Children’s Hospital, a leading pediatric referral center in Senegal. The hospital is located approximately 3 kilometers from the town of Diamniadio, along the national highway toward Thiès.
Inclusion and Exclusion Criteria
Inclusion criteria: All children and adolescents diagnosed with sickle cell disease (SCD) who received care at Diamniadio Children’s Hospital during the study period were included.
Exclusion criteria: Patients without a diagnosis of SCD who were treated at the hospital during the same period were excluded from the analysis.
Data Collection
Patient data were extracted from hospital registries and compiled into a Microsoft Excel spreadsheet. The variables collected included:
Results
- Demographic information (age and sex)
- Hemoglobin electrophoresis profile
- Reason for consultation
- Identified pathogens
- Results of antibiotic susceptibility testing.
Among the 240 patients with sickle cell disease included in this study, 56 (23.3%) presented with infectious complications. Analysis of the socio-demographic characteristics of these 56 patients revealed a slight predominance of females (55.36%) over males (44.64%). The majority of infections occurred in children under 10 years of age, with 53.6% of cases in the 0–5years age group and 35.7% in the 5–10year age group. Only 10.7% of cases were observed in patients older than 10 years (Table 1)
Septicemia was the most common infection, representing 37% of cases, followed by osteomyelitis (25%) and bronchopneumonia (20%).
Less frequent infections included urinary tract infections (9%), arthritis (5%), and myositis (4%). This pattern underscores the predominance of invasive bacterial infections particularly bloodstream and bone infections among children with sickle cell disease (figure 1)
The analysis of pathogens isolated from patients with infectious complications revealed that Salmonella spp. was the most prevalent, responsible for 29% of cases. This was followed by Staphylococcus aureus (16%), Escherichia coli and Streptococcus pneumoniae (each 14%). Less frequently identified pathogens included Citrobacter freundii (11%), Proteus mirabilis (9%), and Klebsiella oxytoca (7%) (Figure 2).
Antibiotic susceptibility testing of the isolated Salmonella spp. strains revealed complete sensitivity to all antibiotics evaluated including Amoxicillin, Amoxicillin-Clavulanic Acid, Ertapenem, Cefoxitin, Aztreonam, Colistin, Netilmicin, Norfloxacin, and Cotrimoxazole (figure 3).
Discussion
Among the 240 patients with sickle cell disease (SCD) included in this study, 56 (23.3%) presented with infectious complications. This prevalence aligns with global data indicating that infection remains a leading cause of morbidity and mortality in children with SCD, particularly in Senegal [7].
The socio-demographic analysis revealed a slight predominance of females (55.36%) over males (44.64%) among infected patients. While gender differences in infection susceptibility are not consistently reported in the literature, some studies suggest that hormonal and immunological factors may influence disease expression [8]. More notably, the age distribution showed that 89.3% of infections occurred in children under 10 years, with the highest burden in the 0–5year age group (53.6%). This is consistent with evidence that functional asplenia develops early in life, often before the age of five, leaving young children particularly vulnerable to encapsulated bacteria such as Streptococcus pneumoniae, Haemophilus influenzae, and Salmonella spp. [9]. In this study, septicemia emerged as the most common infectious complication among children with sickle cell disease (SCD), accounting for 37% of cases, followed by osteomyelitis (25%) and bronchopneumonia (20%). These findings reflect the well-documented vulnerability of children with SCD to invasive bacterial infections, particularly those affecting the bloodstream and bones [10, 7]. The high prevalence of septicemia is consistent with the pathophysiology of SCD, where functional asplenia often occurring within the first few years of life compromises the immune system’s ability to clear encapsulated bacteria such as Streptococcus pneumoniae, Haemophilus influenzae, and Salmonella spp. [11,9].
In low-resource settings, where access to early diagnosis, vaccination, and antibiotics may be limited, septicemia remains a leading cause of mortality [12]. With a prevalence of 29%, Salmonella emerges as the leading pathogen in pediatric patients with sickle cell disease. This aligns with findings from a large European multicenter study (2014–2019), which showed that Salmonella spp. was the most frequently isolated pathogen in IBIs among children with SCD, particularly in osteomyelitis and primary bacteremia [10]. Salmonella is especially problematic in SCD due to bone infarctions, which create a favorable environment for bacterial colonization and infection [13].
The observation that Salmonella spp. isolates demonstrated complete sensitivity to a broad spectrum of antibiotics is a significant and encouraging finding, particularly in the context of rising global antimicrobial resistance (AMR). Nevertheless, global trends indicate increasing resistance in Salmonella, particularly to fluoroquinolones and third-generation cephalosporins [14].
Conclusion
These findings underscore the critical need for routine screening of Salmonella infections, particularly in young children living with sickle cell disease. The predominance of invasive infections and the central role of Salmonella call for enhanced infection prevention strategies, including vaccination, prophylactic antibiotic use, and early intervention protocols. The observed antibiotic susceptibility of Salmonella supports the continued use of current treatment regimens, while also highlighting the importance of sustained antimicrobial stewardship to preserve the long-term effectiveness of available therapies.
Acknowledgements
We thank all of people who have participated in this research.
Funding Information
There has been no funding for this research.
Ethics Approval and Consent to Participate
Not relevant.
Conflict of Interest
There has been no conflict of interest.
References
- Modell B, Darlison M (2008) Global epidemiology of haemoglobin disorders and derived service indicators. Bull World Health Organ. 86: 480-7.
- Seck M, Dabo MA, Bousso ES, Keita M, Touré SA, et al. (2024) Homozygous Sickle Cell Disease after Age of 40: Follow-Up of a Cohort of 209 Patients in Senegal, West Africa. Adv Hematol. 2024: 501577.
- WHO. Child mortality and causes of death. Available https://www.who.int/data/gho/data/themes/topics/topic-details/GHO/child-mortality-and-causes-of-death. Consulté le 02/09/2025
- WHO. Child mortality (under 5 years). Available https://www.who.int/news-room/fact-sheets/detail/child-mortality-under-5-years. Consulté le 02/09/2025)
- Ngoungou EB, Bisvigou UJ, Engohang-Ndong J, Anguezomo V, Sydney MN, et al. (2025) Determinants of immunisation in children with sickle cell disease in Libreville. J Public Health Afr. 16: 663.
- Kane A, Mbaye A, Sow A, Sy B, Thiongane A, et al. (2025) Infection and Impact of Prophylaxis in Sickle Cell Children: A Cross-Sectional Study of 327 Sickle Cell Children Admitted in Emergency Department of Albert Royer Children Hospital in Dakar, Senegal. Open Journal of Pediatrics, 15: 177-88.
- Ochocinski D, Dalal M, Black LV, Carr S, Lew J, et al. (2020) Life-Threatening Infectious Complications in Sickle Cell Disease: A Concise Narrative Review. Front Pediatr. 8: 38.
- Masese RV, Bulgin D, Knisely MR, Preiss L, Stevenson E, et al. (2021) Sickle Cell Disease Implementation Consortium. Sex-based differences in the manifestations and complications of sickle cell disease: Report from the Sickle Cell Disease Implementation Consortium. PLoS One. 16: e0258638.
- Scourfield LEA, Nardo-Marino A, Williams TN, Rees DC (2025) Infections in sickle cell disease. Haematologica. 110: 546-61.
- Gaschignard J, Koehl B, Rees DC, Rincón-López E, Vanderfaeillie A, et al. (2023) BACT-SPRING study group. Invasive Bacterial Infections in Children With Sickle Cell Disease: 2014-2019. Pediatrics. 152: e2022061061.
- CDC. Complications of SCD: Infection. Available https://www.cdc.gov/sickle-cell/complications/complications-of-scd-infection.html. Consulté le 01/09/2025
- WHO. Sickle-cell disease. Available https://www.who.int/news-room/fact-sheets/detail/sickle-cell-disease. Consulté le 01/09/2025
- Brent AJ, Oundo JO (2020) Salmonella infections in children with sickle cell disease. Journal of Infection in Developing Countries. 14: 1141-6.
- Leshchiner D, Rosconi F, Sundaresh B et al. (2022) A genome-wide atlas of antibiotic susceptibility targets and pathways to tolerance. Nat Commun 13: 3165.
Article Information
Research Article
Received Date: September 03, 2025
Accepted Date: September 16, 2025
Published Date: September 20, 2025
Sickle Cell and Salmonella: A Hidden Health Threat for Senegalese Children
Volume 1 | Issue 2
Citation
Assane Dieng, Abou Aw, Niokhor Ngobe Sene, Awa Ba Diallo, Gora Lo, et al. (2025) Sickle Cell and Salmonella: A Hidden Health Threat for Senegalese Children. J Microbiol and Bacteriol Res 1: 202
Copyright
©2025 Assane Dieng. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

doi: jmbr.2025.1.202

