
Research Article
Volume-1 Issue-2, 2025
Comparative Studies on the Yield of Biodiesel from Different Sourced Waste Cooking Oils Using Homogenous KOH Catalysis
Received Date: November 13, 2025
Accepted Date: December 03, 2025
Published Date: December 05, 2025
Journal Information
Switch to Full Text Menu
Abstract
In this study, transesterification method is used to produce biodiesel from different waste cooking oil (ghee oil, soyabean oil, sunflower oil and rice bran oil). Homogenous base catalyst, Potassium hydroxide (KOH) is used in presence of methanol. The comparison and effect of different parameters like reaction temperature, time of reaction, amount of catalyst, and molar ratio of methanol to oil on the yield of biodiesel are studied for different waste cooking oils (WCO). Results revealed that 98.1±1 % yield is achieved for ghee oil with 55oC, 30 mins of reaction time and 0.98 g of catalyst. Maximum yields of 98.7±1%, 97.8±1.8% and 95.9±1.8%, were achieved at 60oC, and 0.98 g of catalyst for cooked sunflower oil, rice bran oil and soyabean oil respectively. GC-MS was also carried out to identify the composition of the methyl ester present in the synthesized biodiesel. Attempt is made to develop a correlation among the yield and the experimental parameters. The calculated values of the yield obtained through the developed correlation is also compared against the experimentally observed yield-values which is found to have a good agreement with standard deviation and mean deviation of 5.66 and -0.05 respectively.
Key words
Keywords: Waste Cooking Oil, KOH Catalyst, Transesterification, Biodiesel, Optimum Condition
| Item | SV | AV | M | Compositions |
| Ghee oil | 192 | 0.75 | 864 | 98.9% lipids, 0.3% water and less than 0.9% nonfat solids. |
| Rice bran oil | 200 | 1.08 | 846 | 30% linoleic acid, 44% oleic and about 23% saturated fatty acids. |
| Sunflower oil | 198 | 1.28 | 855 | 14–43% oleic and 44–75% linoleic acids and 15% saturated fatty acids. |
| Soyabean oil | 175 | 0.56 | 964 | palmitic acid (16:0), stearic acid (18:0), oleic acid (18:1), linoleic acid (18:2), and linolenic acid (18:3) are in 10%, 4%, 18%, 55%, and 13% respectively. |
| Sample number | Temperature (T,°C) | Vol. of WCO (ml) | Time (t,min.) | Catalyst KOH (W, gm) | Methanol (ml) | Waste Cooking oil |
| 1 | 50 | 100 | 30 | 0.98 | 20 | Ghee Oil |
| 2 | 55 | 100 | 30 | 0.98 | 20 | Ghee Oil |
| 3 | 60 | 100 | 30 | 0.98 | 20 | Ghee Oil |
| 4 | 65 | 100 | 30 | 0.98 | 20 | Ghee Oil |
| 5 | 60 | 100 | 25 | 0.98 | 20 | Ghee Oil |
| 6 | 60 | 100 | 35 | 0.98 | 20 | Ghee Oil |
| 7 | 60 | 100 | 40 | 0.98 | 20 | Ghee Oil |
| 8 | 60 | 100 | 30 | 0.9 | 20 | Ghee Oil |
| 9 | 60 | 100 | 30 | 1.1 | 20 | Ghee Oil |
| 10 | 60 | 100 | 30 | 1.15 | 20 | Ghee Oil |
| 11 | 60 | 100 | 30 | 0.98 | 10 | Ghee Oil |
| 12 | 60 | 100 | 30 | 0.98 | 15 | Ghee Oil |
| 13 | 60 | 100 | 30 | 0.98 | 25 | Ghee Oil |
| 14 | 60 | 100 | 30 | 0.98 | 20 | Rice Bran oil |
| 15 | 60 | 100 | 30 | 0.98 | 20 | Sunflower oil |
| 16 | 60 | 100 | 30 | 0.98 | 20 | Soyabean oil |
| Sample Name | Retention time | Identified methyl ester compounds | Percentage |
| Waste Ghee oil | 10.125 | Hexadecanoic acid | 0.1093 |
| 19.229 | 9,12- Octadecadienoic acid | 0.5378 | |
| 19.706 | 9- Octadecenoic acid | 0.1283 | |
| 19.722 | 11-Octadecenoic acid | 0.0258 | |
| 19.753 | 6-Octadecenoic acid | 0.1988 | |
| Waste Ghee oil (55⁰C, 30 min, 20 ml) | 19.198 | 9,12-Octadecadienoic acid | 1 |
| Waste Ghee oil | 10.133 | Hexadecanoic acid | 0.0944 |
| 19.291 | 9,12-Octadecadienoic acid | 0.5336 | |
| 19.789 | 11-Octadecenoic acid | 0.3452 | |
| 22.064 | Methyl stearate | 0.0152 | |
| 22.082 | Methyl stearate | 0.0176 |
 |
| Figure 1: (a) Waste cooking oil, (b) Removal of impurities and inorganic materials, (c) Moisture removal from WCO and (d) Transesterification reaction of WCO with catalyst. |
 |
| Figure 2: (a) Reaction for conversion of WCO to Biodiesel, (b) Biodiesel formation and (c) Separation of biodiesel from glycerol and impurities using separation funnel. |
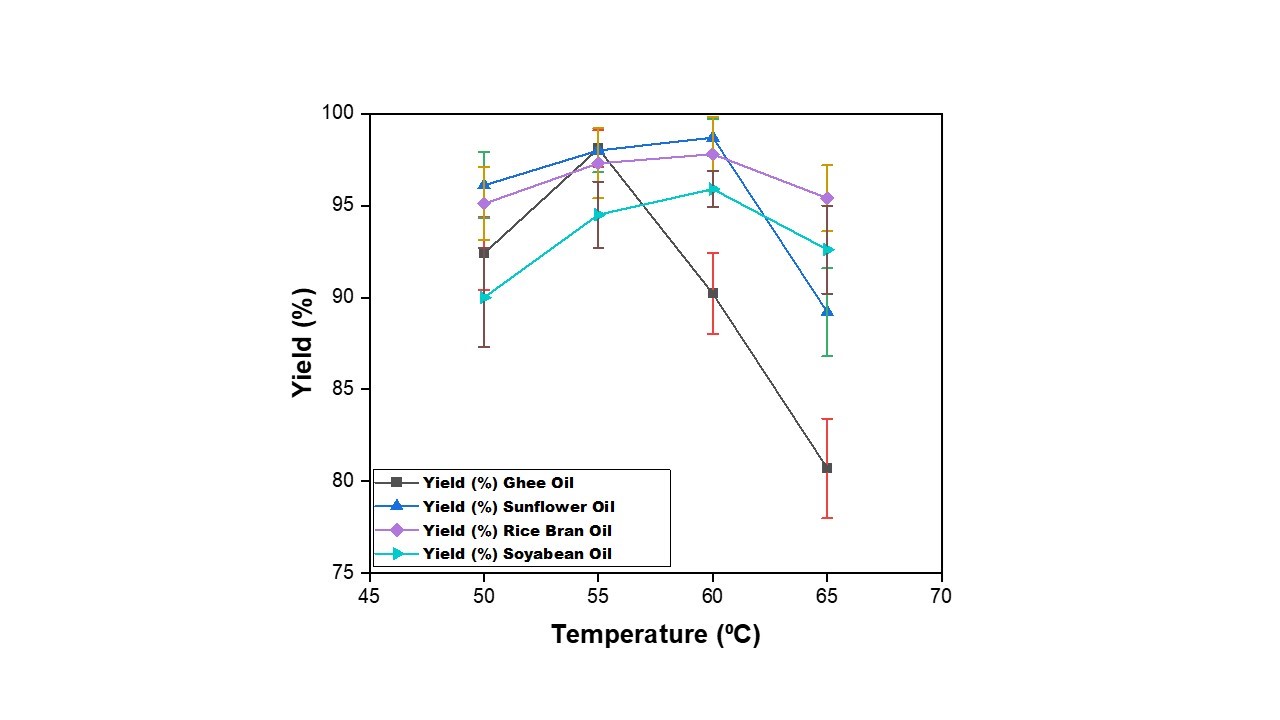 |
| Figure 3: Effect of Temperature on production of biodiesel from waste cooking oil with catalyst = 0.98g, Methanol= 20 ml, and Time= 30 min |
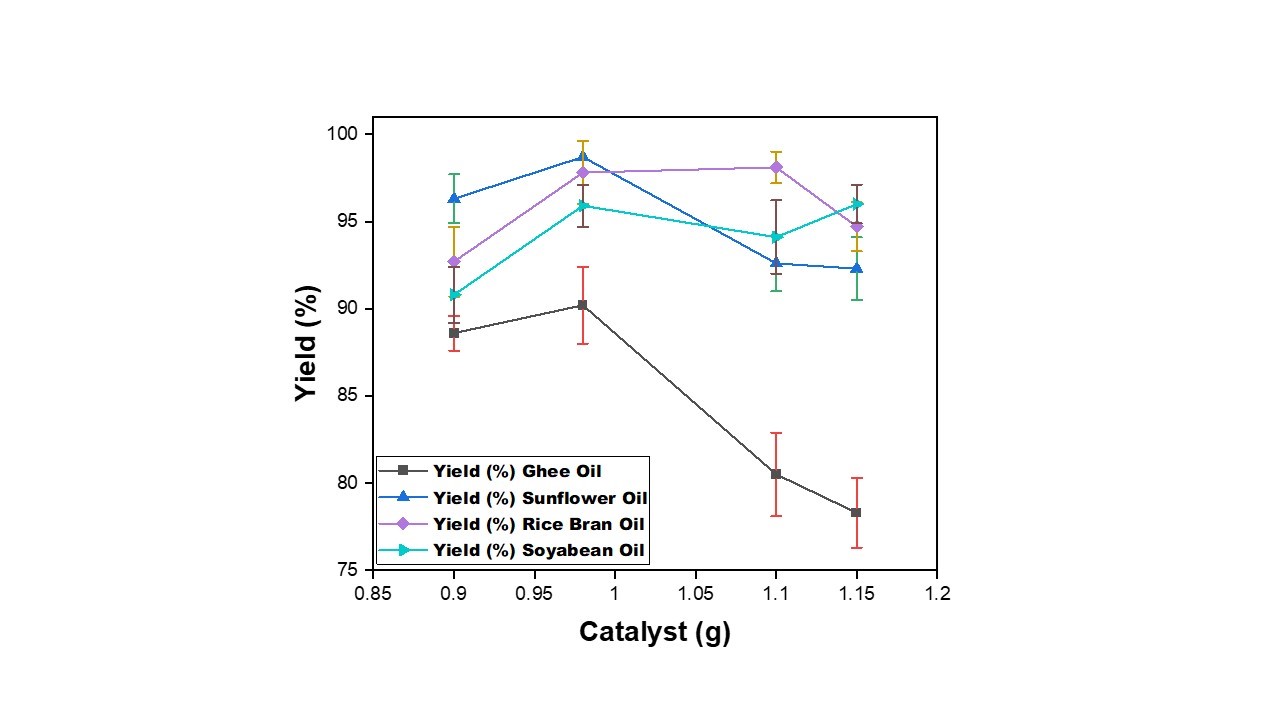 |
| Figure 4: Effect of Catalyst amount on production of biodiesel from waste cooking oil with Temperature = 60⁰C, Methanol=20 ml, Time= 30 min |
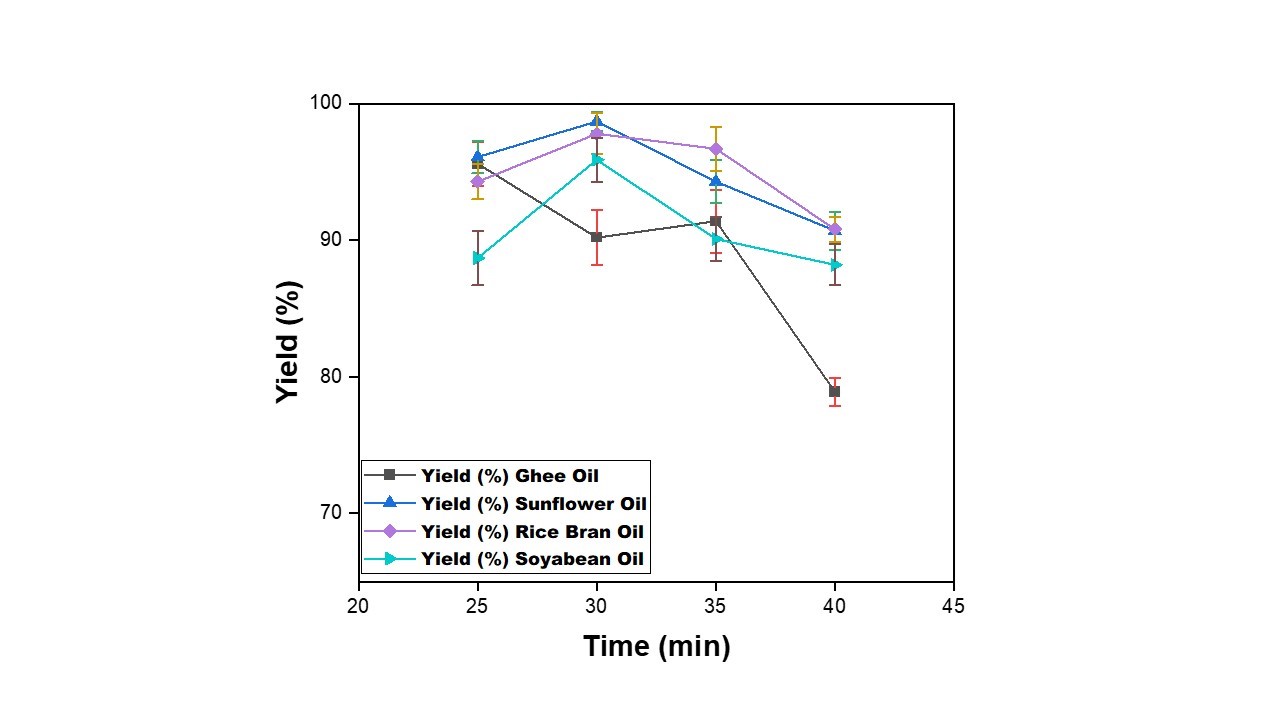 |
| Figure 5: Effect of reaction time on yield of biodiesel from waste cooking oil, Temperature =60⁰C, Methanol=20 ml, Catalyst=0.98 g |
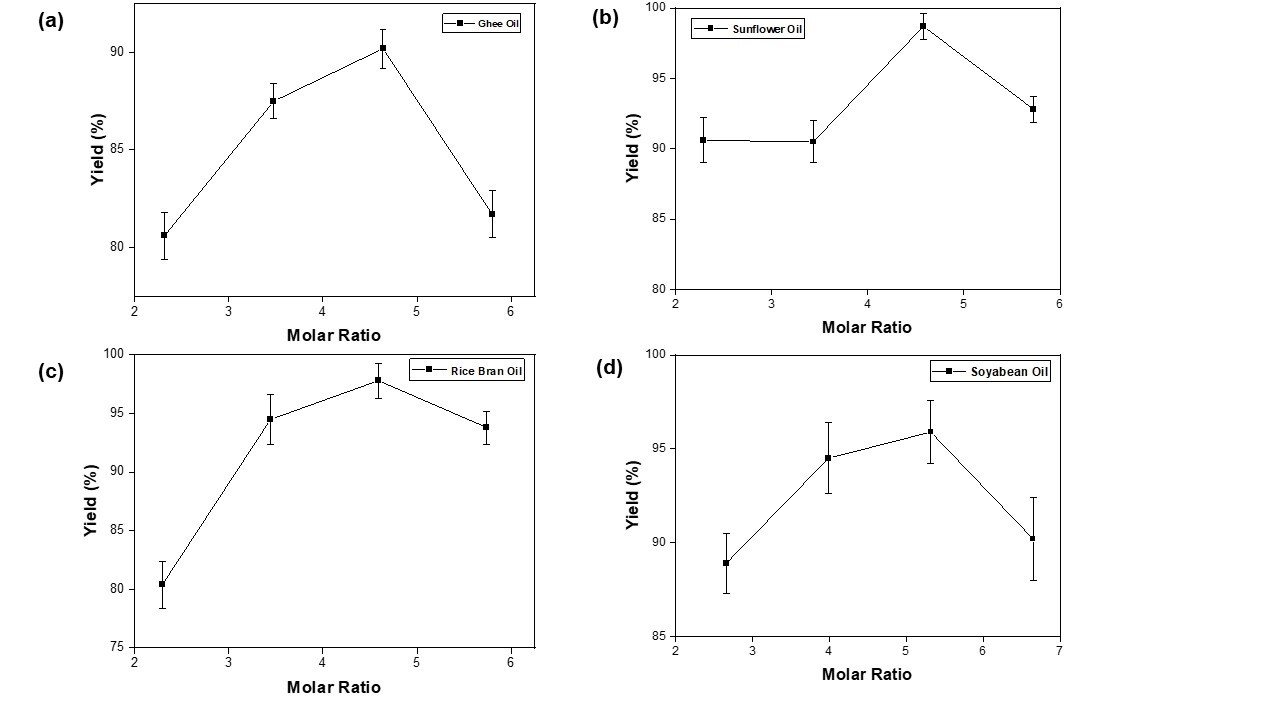 |
| Figure 6: Effect of molar ratio (methanol/WCO) on yield of biodiesel from waste cooking oil with Temperature =60⁰C, Reaction Time = 30 min, Catalyst=0.98 g (a) waste ghee oil, (b) waste sunflower oil, (c) waste rice bran oil and (d) waste soyabean oil. |
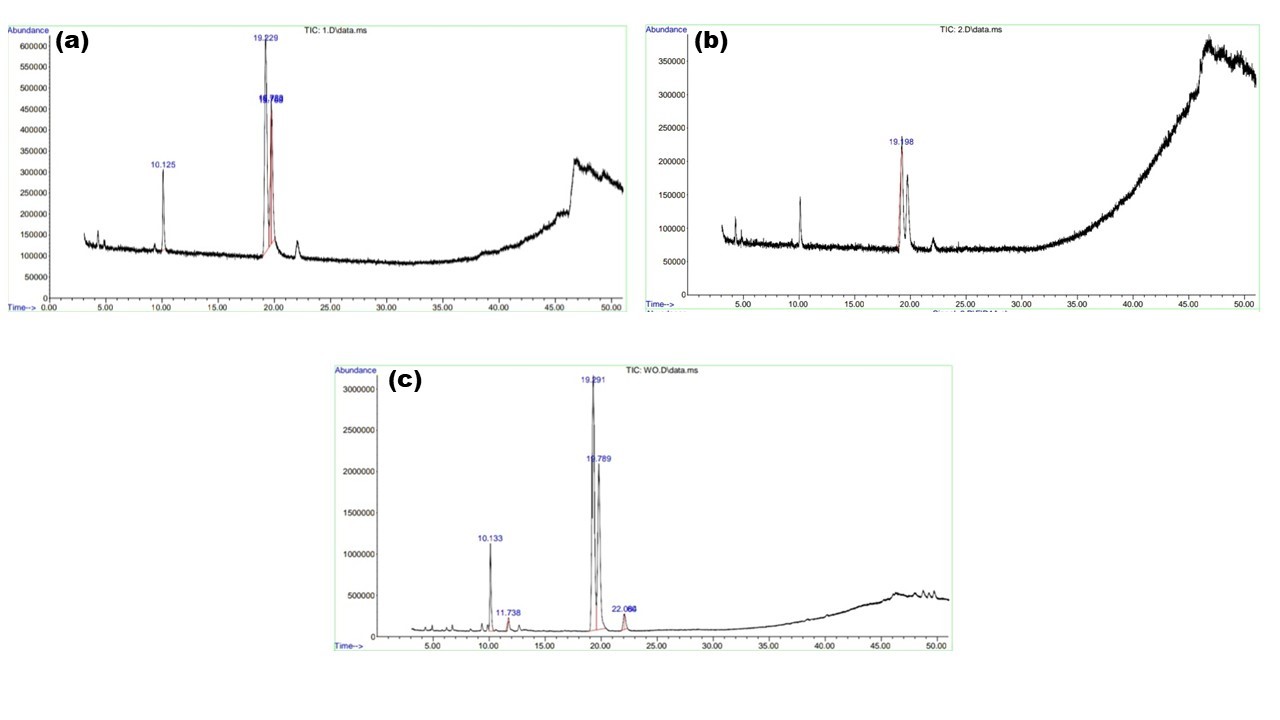 |
| Figure 7: GC-MS curve for biodiesel produced from waste ghee oil with (a) 50⁰C, 30 min, 20 ml, (b) 55⁰C, 30 min, 20 ml, and (c) 60⁰C, 30 min, 20 ml. |
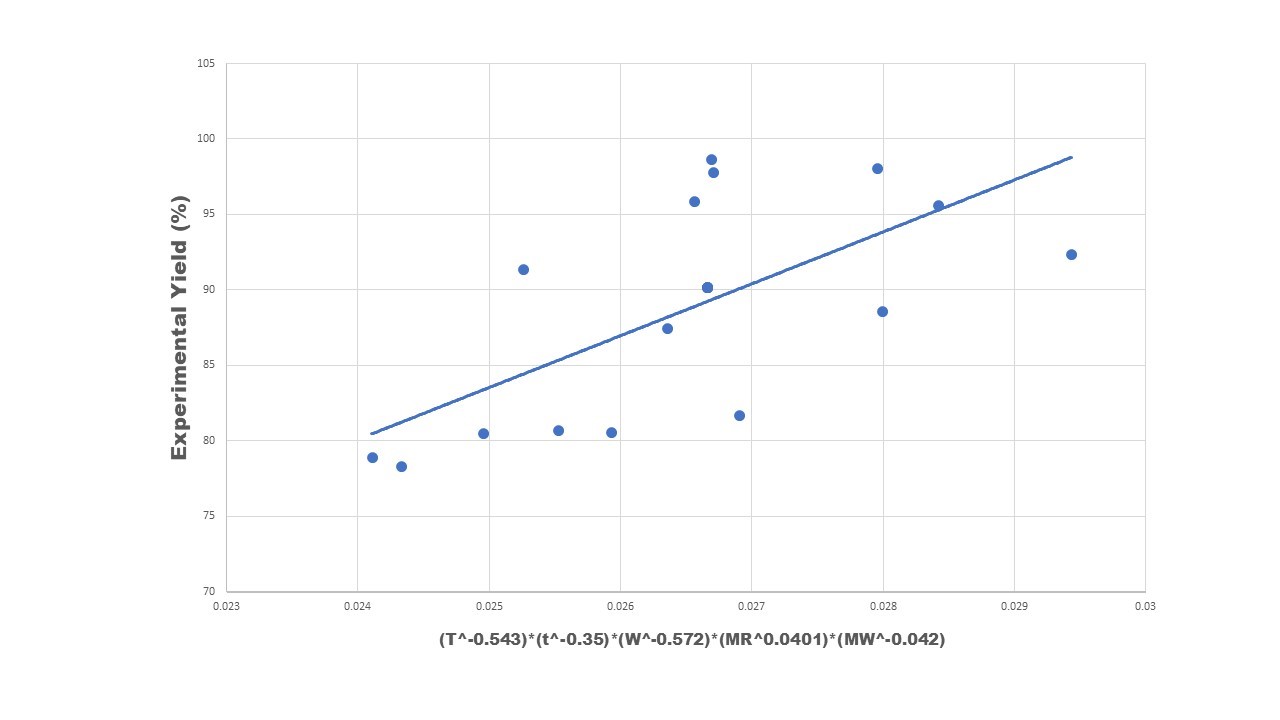 |
| Figure 8: Correlation plot between the experimental yield and different system parameters |
Introduction
Diesels are produced by fractional distillation of fossil fuel, crude oil. Fossil fuel being non-renewable in nature is getting depleted from time to time because of its high usage. India is one of the fast-growing economies which focuses on infrastructure, manufacture and transportation. The reasons for finding alternative fuels to replace crude oil is increasing price of oil products, and greenhouse gas emissions leading to environmental issues. Mainly there are two reasons for constant increase in oil prices. One is increased energy consumptions and the other one is depletion of petroleum reserves. In addition to these reasons, sustainability and environmental issues are also main problems that 21st century is facing. Conventional fuels pollute air by releasing Sulphur dioxide, Carbon dioxide, other gases and particulate matters. Therefore, it is essential to develop renewable energy to face these challenges in the energy sectors [1, 2]. In India, reported oil consumption in 2018 was almost 35 million tons of oil equivalent out of which 70% are fulfilled by import [3] which costs about 800,000 million rupees per year.
Biodiesel can be used as an alternate for fossil fuel such as petro-diesel. According to ASTM biodiesel are mono-alkyl esters of long-chain fatty acids derived from bio-based materials such as animal fat and vegetable oil [4]. It has many benefits compared to petroleum diesel like reduced greenhouse gas emissions, negligible SOx emissions, low PM-pollutants, low toxicity and bio degradability. Different feed stocks with lipid sources like edible oils, non-edible oils, microalgae, animal fat and waste cooking oil are used for synthesis of biodiesel [5]. Biodiesels produced from these sources are found to be superior to petro-diesels in terms of different parameters like exhaust emissions, cetane number, flash point and lubricity characteristics without much differences in the properties of heat of combustion [6].
Refined edible oils are widely used as the primary raw material in the biodiesel industry. But its commercialization is expensive as production costs is high. It has been reported that the cost of feedstock constitutes about 60-80% of the overall cost in the production process [7]. Waste cooking oil (WCO) is obtained from vegetable oils used for cooking or from food processing industries. Waste oil if disposed as it is, leads to soil and water pollution thereby causing human health concerns and also causes disturbance to aquatic eco-system. To overcome this problem, waste cooking oil can be used as a promising feedstock because of its high availability as a waste from household, restaurants and food industry thereby reducing the burden of disposing waste cooking oil. Besides this, cost, availability, government support and performance as a fuel [8] make it an excellent feedstock to develop an alternate fuel source. But the biodiesel produced from WCO should be effective based on its derived performance for different parameters like titre, FFA content, moisture content, calorific content, and other impurities [9]. Many researchers [10] have successfully converted used waste cooking oil into biodiesel. Vegetable oil are saturated hydrocarbons which are made of glycerol and esters of fatty acids. On repeated usage of cooking oil, fatty acid (FFA) content increases due to hydrolysis, oxidation, polymerization and mass transfer between food and vegetable oil. Increase in viscosity and saponification number arises due to oxidation and polymerization reactions [2]. The heat and water used during the cooking increases the triglyceride content due to hydrolysis which further breaks down to Di-glycerides, Mono-glycerides and free fatty acids [2, 11]. The presence of high FFA and saponification number affects the quality of biodiesel.
Biodiesel is mainly synthesized by pyrolysis, micro-emulsion, blending of oils and transesterification. Among all these methods, trans-esterification is the most commonly used as the viscosity of biodiesel generated is decreased in this process and also requires mild operating condition to complete the reaction [12]. Transesterification process is of different types like enzymatic reaction, non-catalysed reaction and chemical catalysed reaction (both acid catalyst and alkali catalyst) [13, 14]. It is observed that chemical catalysed reaction has less reaction time and less operating cost as compared to enzyme catalysts [1]. Heterogeneous chemical catalysts have reported for transesterification of FFA to biodiesel for its easy separation and reusability. But this catalyst requires extreme temperature, diffusion limitation, long reaction time to achieve the desired yield, complex and expensive route and high cost of production of catalyst [15].
Basic catalysts are used in wide range due to its low cost, high reaction rate, mild temperature range and also remove the problem of corrosion that are encountered while using acid-based catalyst [5]. Different studies are reported on synthesis of biodiesel by using KOH as catalyst. It was observed that methanol was used as medium for most of the reactions because of its low cost and easy availability. Studies were reported on synthesis of biodiesel from WCO using methanol and sodium hydroxide as catalyst and further optimized the reaction conditions to achieve an efficiency of 89.8% [16]. Comparison of yield using different catalyst was carried out and found out that biodiesel production yield of 99% is achieved using 1% KOH as catalyst in presence of methanol at 1:6 ratio from waste sunflower cooking oil and pure sunflower cooking oil [17]. In the above studies, biodiesel was produced from single oil and its waste derivatives and no comparison has been made for various other WCO. Alkali-based transesterification of WCO to produce biodiesel in presence of methanol. The experiment resulted yield of 88-90% for 0.75wt% KOH and 7:1 methanol/oil ratio. Besides this, they have also studied the physical properties of the blend [18]. Biodiesel was produced from WCO using sodium hydroxide and methanol where the yield achieved was 90% at 30⁰C and 6:1 alcohol/oil ratio [19]. In this present study, yield of biodiesel and comparison are planned to be studied using different waste cooking oils derived from animal fat and plant derivatives with KOH- catalyst. Besides this, different parameters such as reaction time, methanol to oil ratio, temperature and catalyst concentration are also planned to be studied to optimize the biodiesel reaction. Further, a correlation is thought to be developed to predict the effect of different experimental parameters on the yield of biodiesel.
Materials and Methods
Materials
Waste cooking oils from different sources viz. ghee oil (animal fat), rice bran oil, soya bean oil and sunflower oil were collected from kitchens of local halls of residence and restaurants. Potassium hydroxide (KOH), methanol of analytical grade was used for catalyst and solvent, respectively for transesterification process.
Transesterification of Waste Cooking Oil
The saponification value (SV) and acid value (AV) of the oil (WCO) samples are provided in Table 1[20-22]. The molecular weight of the waste cooking oils was calculated from the equation
M= 56.1*1000*3/(SV-AV) (1)
Experiment was performed as described earlier [23]. Prior to the experiment, the waste cooking oil samples were filtered to remove the inorganic materials and the impurities present in the oil as represented in Figure 1(a)-(b). The filtered oils were then heated at 60°C for 30 to 60 minutes to remove the moisture present as shown in Figure 1(c). Different solution of catalysts was prepared by adding different mass of KOH with methanol. The prepared catalyst solution was added in waste cooking oil and was stirred in an agitator till the solution turned murky. The above prepared solution was then transferred to a round bottomed flask and kept on heating mantle for heating at different temperatures and time period as indicated in Figure 1(d).
After the reaction, the sample was transferred to a separation-funnel where the sample was kept for 8 h for proper separation of glycerol and biodiesel. The solution containing glycerol, soap and impurities collected at the bottom were then removed from the solution as shown in Figure 2(c). The biodiesel obtained from top was then washed with warm water and dried to remove residual catalyst. The variation in different parameters for synthesis of biodiesel are summarized in Table 2.
After the reaction, the sample was transferred to a separation-funnel where the sample was kept for 8 h for proper separation of glycerol and biodiesel. The solution containing glycerol, soap and impurities collected at the bottom were then removed from the solution as shown in Figure 2(c). The biodiesel obtained from top was then washed with warm water and dried to remove residual catalyst. The variation in different parameters for synthesis of biodiesel are summarized in Table 2.
Results and Discussions
Effect of Temperature
To study the effect the temperature on the yield, the reaction was repeated at various temperatures as discussed in Table-2. Effect of temperature on yield of biodiesel has been compared for four different oil samples as shown in Figure 3. It is observed that yield of biodiesel initially increases for ghee oil, rice bran, soybean oil and sunflower oil with the increase in temperature from 50 to 55oC. The biodiesel yield achieved is in a range from 90-95%. Further, with the increase in temperature up to 60⁰C, the yield of ghee oil reduces to around 90.2% whereas an increase trend is observed for other oils with the achieved yield above 95%. The reduction in the yield in ghee oil may be due to formation of soap by saponification. With further increase in temperature above 60oC the biodiesel yield is found to decrease continuously for all oils which may be due to evaporation of methanol [24]. So, for studying the effects of other parameters on the yield of biodiesel, 60oC was taken as the optimum temperature.
As per literature [29], standard value of temperature at which transesterification reaction with alkali-catalyst takes place is 60 oC. However, depending upon the oil-source, catalyst used and different degrees of conversion, transesterification reaction temperature ranges from 25 to 120oC. That is why in the present work, operating temperature is maintained at 60 o C. To study the effect of temperature on yield of biodiesel, temperature is varied around 60oC (50-65oC).
Effect of Quantity of Catalyst
The presence of catalyst helps in accelerating the reaction. But excess use of the catalyst may have an adverse impact on the yield thereby increases the cost. In order to optimize the utility of the catalyst, amount of KOH-catalyst was varied from 0.9 to 1.15 g. The experiment was carried out to study the effect of catalyst on the yield of biodiesel for different oils as shown in Figure 4.
It is observed that with increase in the amount of catalyst, the yield of biodiesel for all the WCO increases in the range of 90% to 95% with increase in amount catalyst from 0.9 to 0.98 g. This may be due to the increase in the availability of active sites for the reaction to take place. It is further observed that with increase in the amount of catalyst from 0.98 to 1.1g yield% is decreasing for ghee oil and sunflower oil while a little increase is seen in the yield for rice bran and soyabean oil. This may be due to the formation of soap by saponification of fatty acids as side reaction [25]. A further increase in catalyst concentration also leads to extra costs, because it is necessary to remove it from the reaction medium at the end of the process [26]. Furthermore, Dorado et al. [26] concluded that the addition of an excessive amount of alkaline catalyst gives rise to the formation of an emulsion, which increases viscosity and leads to the formation of gels. In order to optimize the utility of the catalyst, amount of KOH-catalyst was varied from 0.9 to 1.15 g to keep around 1% by wt. of oil. A further increase in catalyst concentration also leads to extra costs, because it is necessary to remove it from the reaction medium at the end of the process [28]. Furthermore, Dorado et al. [26] concluded that the addition of an excessive amount of alkaline catalyst gives rise to the formation of an emulsion, which increases viscosity and leads to the formation of gels. Alamu et al.[29] found the yield of palm biodiesel was 95.8% under 100 g palm oil, 20.0% ethanol (wt% palm oil), 1.0% NaOH catalyst [21], 60 °C reaction temperature and 90 min reaction time.
Effect of Reaction Time
Effect of time on the yield of biodiesel was also studied for different waste cooking oils by varying time of reaction (viz. 25, 30, 35 and 40 mins) as shown in Figure 5. It is observed that biodiesel yield increases with increase in reaction time for all WCO till 30 min except for ghee oil. With further increase in reaction time beyond 30 mins a decline in yield is observed for all WCO. This may due to the commencement of saponification reaction as side reaction and also due to the reversible nature of reaction with increased time of reaction [27]. The maximum yield of 95.6% at 25 mins for ghee oil while 98.7%, 97.8% and 95.9% at 30 min are achieved for sunflower, rice bran and soyabean oils, respectively.
As per literature [8], the maximum ester conversion has been found to be attained within 90 min. In the beginning the reaction is slow due to mixing and dispersal of alcohol and oil, then after some time the reaction proceeds very fast.
Mathiyazhagan & Ganapathi (2011) and Jagadale & Jugulkar (2012) and many more researchers have reported that maximum yield of biodiesel is achieved within 90-minutes. It was also reported by Jagadale & Jugulkar (2012) for Chicken Fat Based Biodiesel that there was a great jump from 30 minute to 60 minute which indicated that the reaction was very rapid in the beginning. As waste cooking oil from plant-sourced material was considered in the present work, time range was considered 25 - 40 min less than the range of 30 – 60 minutes.
Effect of Alcohol and WCO Ratio
Effect of methanol to oil molar ratio was studied for different waste cooking oils by varying methanol volume (i.e. 10, 15, 20 and 25 ml). Molar ratio was calculated as 2.36, 3.54, 4.72 and 5.90 respectively. The effect of molar ratio on yield of biodiesel is indicated in Figure 6 (a-d). It is observed that biodiesel yield increases with the increase in molar ratio from around 2.36 to 4.72 for all WCO samples. The highest yield of 90.2%, 98.7%, 97.8% and 95.9% are achieved for ghee oil, sunflower oil, rice bran oil and soya bean oil respectively at molar ratio of 4.64, 4.58, 4.59 and 5.32. This may be due to the presence of excess methanol for which the transesterification reaction moves in forward direction thereby increasing the yield of biodiesel. With further increase in the molar ratio up to 5.90, the yield is found to decrease drastically which may be because of the dilution of the catalyst and mixing of glycerol with the biodiesel[28]. Alamu et al.[29] found the yield of palm biodiesel was 95.8% under 100 g palm oil, 20.0% ethanol (wt% palm oil), 1.0% NaOH catalyst, 60 °C reaction temperature and 90 min reaction time. As per literature [29], for alkaline catalyzed transesterification process, 6 moles of alcohol are required per mole of triglyceride in turn per mole of oil (6:1 molar ratio) for maximum yield [28]. As per literature [29] ethanol amount around 20% by weight of oil yields That is why attempt was made to keep alcohol to oil molar ratio about 6 (5.9) in the present work. Again, to check the effect of molar ratio on yield of biodiesel, molar ratio was varied as 2.36, 3.54, 4.72 and 5.90 keeping volume% around 20. Further, to compare the consumption of alcohol on yield of biodiesel, oil volume was kept constant at 100ml for different oils in the transesterification process.
GC-MS Analysis
Biodiesel is a mixture of various types of methyl esters. To identify the percentage amount and composition of the biodiesel, GC-MS analysis was performed on biodiesel produced from waste ghee oil at 50⁰C, 55⁰C, 60⁰C. The peaks of GC-MS are shown in Figure 7. It is observed that multiple peaks are seen at retention time for biodiesel yield from waste ghee oil at 50⁰C which are represented in Table 3. From the table it is found out that the biodiesel has both saturated and unsaturated fatty acids of methyl esters. Waste ghee oil has a maximum percentage of 9,12- Octadecadienoic acid, followed by 6-Octadecenoic acid and Hexadecanoic acids.
Analysis on Biodiesel Yield
Yield percentage of biodiesel from waste cooking oil depends on many factors like temperature (T), reaction time (t), alcohol to oil molar ratio (MR), type of waste cooking oil i.e. molecular weight of oil (MW), catalyst concentration (W) and many more. Attempt is made to develop a correlation relating the biodiesel yield with these system parameters which is shown below.
Yield % = 3.7×103 [T -0.558 × t -0.36 × W -0.588 × MR 0.041 × MW-0.043] (2)
Where, T- Temperature, t- reaction time, W- amount of catalyst, MR- methanol to oil ratio, MW- molecular weight of waste cooking oil.
The correlation plot is shown in Figure 8. Negative power is observed for temperature, time, amount of catalyst and molecular weight. A negative yield is observed with high temperature due to the evaporation of methanol. High catalyst content reduces the yield as it gets mixed with the solvent and also accelerate side reaction of saponification. Molecular weight has a negative impact on yield as compared to other parameters as with high molecular weight the presence of free fatty acids (FFA) increases. These FFA reduces the yield of biodiesel as it promotes the saponification reaction than that of transesterification reaction. Methanol to oil molar ratio is observed to show a positive impact on biodiesel yield. This may be due to the fact that with increase in the volume of methanol, the transesterification reaction moves in forward direction and thus increases the yield of biodiesel. But, the quantity of methanol is subjected to certain limitation as excess methanol can lead to reversible reaction and reduces the biodiesel yield. The standard deviation and mean deviation of 5.66 and -0.05 respectively was achieved resulting in good fitting of data.
Conclusions
Biodiesel was synthesized from four different waste cooking oil collected from hostel and restaurants. Base catalyst KOH was used as the catalyst for synthesis of biodiesel for its low cost and high catalytic activity. Effect of different parameters on biodiesel yield were studied. A maximum yield of 98.1±1% was achieved for ghee oil at 55⁰C. Biodiesel yield of 98.7±0.9%, 97.8±1.2% and 95.9±1.8% were achieved for waste sunflower, rice bran and soyabean oil respectively at temperature of 60⁰C. The optimum conditions achieved for maximum yield of biodiesel from the four-waste cooking oil are summarized as 0.98g of KOH, temperature of 60⁰C, reaction time of 30 minutes and methanol to oil ratio of 4.72. Excess of catalyst showed a negative yield of biodiesel due to the saponification side reaction. Low methanol to oil molar ratio hinders the forward reaction whereas with higher molar ratio dilution of glycerol takes place and thus separation of biodiesel gets difficult. GC-MS confirmed the formation of ndifferent types of methyl esters out of which the methyl ester 9,12-Octadecadienoic acid was the predominant compound for biodiesel obtained from waste ghee oil and thus confirmed the formation of biodiesel. A correlation was developed to study the effect of different parameters on the experimental yield. The experimental yield has shown a negative effect for temperature, reaction time, amount of catalyst and molecular weight of oil whereas for methanol to oil molar ratio it has a positive effect. The calculated values of biodiesel yield through the derived correlation is found to have a good agreement with the experimentally observed biodiesel yield having standard deviation and mean deviation of 5.68 and -0.15 respectively. This implies that the developed correlation can be used over a wide range of parameters and can provide a basis for estimation of biodiesel yield in the commercial scale.
References
- Rahadianti ES, Yerizam, Martha (2018) Biodiesel Production from Waste Cooking Oil Indones. J. Fundamental Appl. Chem. 3: 77.
- Raqeeb MA, Bhargavi R (2015) Biodiesel production from waste cooking oil J. Chem. Pharm. Res. 7: 670.
- Gaur A, Mishra S, Chowdhury S, Baredar P, Verma P (2021) A review on factor affecting biodiesel production from waste cooking oil: An Indian perspective Mater. Today: Proc, 46: 5594.
- Hoekman SK, Broch A, Robbins C, Ceniceros E, Natarajan M (2012) Review of biodiesel composition, properties, and specifications Renew. Sustain. Energy Rev. 16: 143.
- Ferrusca MC, Romero R, Martinez SL, Ramirez-Serrano A, Natividad R (2023) Biodiesel Production from Waste Cooking Oil: A Perspective on Catalytic Processes Processes 11: 1.
- Mahesh SE, Ramanathan A, Begum KMMS, Narayanan A (2015) Biodiesel production from waste cooking oil using KBr impregnated CaO as catalyst Energy Convers. Manag. 91: 442.
- Tesser R, Serio MD, Guida M, Nastasi M, Santacesaria E (2005) Kinetics of oleic acid esterification with methanol in the presence of triglycerides Ind. Eng. Chem. Res 44: 7978.
- Saini RD (2017) Conversion of Waste Cooking Oil to Biodiesel Int. J. Petrol. Sci. Tech. 11
- Yuan W, Yang F (2018) In Taylor and Francis Group Biomass to renewable Energy Processes (2nd edition) 299.
- Gnanaprakasam A, Sivakumar VM, Surendhar A, Thirumarimurugan M, Kannadasan T (2013) Recent Strategy of Biodiesel Production from Waste Cooking Oil and Process Influencing Parameters: A Review J. Energy, 926392.
- Haigh KF, Vladisavljevic GT, Reynolds JC, Nagy Z, Saha B (2014) Kinetics of the pre-treatment of used cooking oil using Novozyme 435 for biodiesel production Chem. Eng. Res. Des. 92: 713.
- Fukuda H, Kondo A, Noda H (2001) Biodiesel fuel production by transesterification of oils J. Biosci. Bioeng. 92: 405.
- Norjannah B, Ong HC, Masjukia HH, Juanb JC, Chong WT (2016) Enzymatic transesterification for biodiesel production: a comprehensive review RSC Adv. 6: 60034.
- Marx S (2016) Glycerol-free biodiesel production through transesterification: a review Fuel Process. Technol. 151: 139.
- Changmai B, Vanlalveni C, Ingle AP, Bhagat R, Rokhum SL (2020) Widely used catalysts in biodiesel production: a review RSC Adv. 10: 41625.
- Meng X, Chen G, Wang Y (2008) Biodiesel production from waste cooking oil via alkali catalyst and its engine test Fuel Process. Technol. 89: 851.
- Hossain ABMS, Boyce AN (2009) Biodiesel production from waste sunflower cooking oil as an environmental recycling process and renewable energy Bulg. J. Agric. Sci. 15: 312.
- Phan AN, Phan TM (2008) Biodiesel production from waste cooking oils Fuel 87: 3490.
- Abubakar HG, Abdulkareem AS, Jimoh A, Agbajelola OD, Okafor JO, et al (2016) .Optimization of biodiesel production from waste cooking oil Energy Sources Part A 38: 2355.
- Prarthana J, Nischitha, Shraddha, Ramu R (2023) Effect of Repeated Heating on Chemical Properties of Selected Edible Plant Oil and its Health Hazards Int. J. Health Allied Sci. 12: 114.
- Deshpande AD, Chavda PD, Kadeval H (2016) Production of Biodiesel Fuel from Waste Soya bean Cooking Oil by Alkali Trans-esterification Process CWE 11: 260.
- Huaping Z, Zongbin W, Yuanxiong C, Ping Z, Shijie D, et al. (2006) Preparation of Biodiesel Catalyzed by Solid Super Base of Calcium Oxide and Its Refining Process Chinese J. Catal. 27: 391.
- Vishnupriya I, Sastry SVAR, Sahoo A (2019) Acid Catalyst of Waste Cooking Oils for Biodiesel Production IJEE 10: 91.
- Abbah EC, Nwandikom GI, Egwuonwu CC, Nwakuba NR (2016) Effect of Reaction Temperature on the Yield of Biodiesel From Neem Seed Oil , AJES 3: 16.
- Agarwal M, Chauhan G, Chaurasia SP, Singh K (2012) Study of catalytic behavior of KOH as homogeneous and heterogeneous catalyst for biodiesel production J. Taiwan Inst. Chem. Eng. 43: 89.
- MP Dorado, E Ballesteros, FJ López, M Mittelbach (2004) Optimization of alkali-catalyzed transesterification of brassica carinata oil for biodiesel production, Energy Fuels, 18: 77-83.
- Erchamo YS, Mamo TT, Workneh GA, Mekonnen YS (2021) Improved biodiesel production from waste cooking oil with mixed methanol-ethanol using enhanced eggshell-derived CaO nano-catalyst Sci. Reports 11: 1.
- Lin YC, Hsu KH, Lin JF (2014) Rapid palm-biodiesel production assisted by a microwave system and sodium methoxide catalyst Fuel 115: 306.
- OJ Alamu, TA Akintola, CC Enweremadu, AE Adeleke (2008) Characterization of palm- kernel oil biodiesel produced through NaOH-catalysed transesterification process, Sci Res Essay, 3: 308-11.
Article Information
Research Article
Received Date: November 13, 2025
Accepted Date: December 03, 2025
Published Date: December 05, 2025
Comparative Studies on the Yield of Biodiesel from Different Sourced Waste Cooking Oils Using Homogenous KOH Catalysis
Volume 1 | Issue 2
Citation
RM Sadini Deshani, Abanti Sahoo, Debasis Nanda, SS Mohapatra (2026) Comparative Studies on the Yield of Biodiesel from Different Sourced Waste Cooking Oils Using Homogenous KOH Catalysis. J Energy Renewable Resour 1: 202
Copyright
©2025 Abanti Sahoo. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

doi: jerr.2025.1.202

